Kannalife Sciences, a subsidiary of Neuropathix, has focused its science on the research and development of a pipeline of next generation breakthrough therapeutics targeting inflammation.
Our lead compound KLS-13019, is a first-in-class, novel molecule that has been validated through IND enabling studies to prevent and reverse chemotherapy induced peripheral neuropathy (CIPN).
Scientific Mission & Purpose
Kannalife Sciences is a pioneer in developing a new class of safe, potent, and selective synthetic analogues of cannabidiol (CBD). The company is dedicated to solving critical unmet needs by advancing innovative, non-opioid, anti-inflammatory, and neuroprotective compounds into clinical development.
Primary Objectives:
- Reducing Opioid Reliance: Decreasing the pain management footprint and dependency on drugs that exacerbate addiction crises.
- Cellular Healthspan Improvement: Targeting mitochondrial dysfunction and neuroinflammation to improve overall cellular healthspan.
- Neurological Legacy: Addressing neuropathic pain, chronic inflammation, opioid addiction, and complications from chemotherapy or aging-related pathways.
Lead Candidate: KLS-13019
KLS-13019 is a globally patented, first-in-class New Chemical Entity (NCE). It is a highly bioavailable (67%) synthetic CBD analogue that is fast-acting and capable of penetrating the blood-brain barrier.
Dual Mechanism of Action (MOA)
KLS-13019 functions through a unique dual mechanism to provide both anti-inflammatory and neuroprotective benefits:
- Anti-Inflammatory (GPR55 Antagonism): It targets and blocks GPR55, a pro-inflammatory receptor. By acting as a GPR55 antagonist, it prevents the priming of NLRP3 inflammasome assembly, which is an early mediator of inflammatory cascades.
- Neuroprotection (Mitochondrial Resilience): It promotes mitochondrial homeostasis by regulating the NCX-1 (Na+/Ca2+ exchanger). This regulation helps maintain the calcium balance necessary for healthy mitochondria, preventing damage caused by reactive oxygen species (ROS).
Therapeutic Pipeline
Kannalife Sciences is advancing a robust pipeline of indications for its lead candidate and secondary compounds.
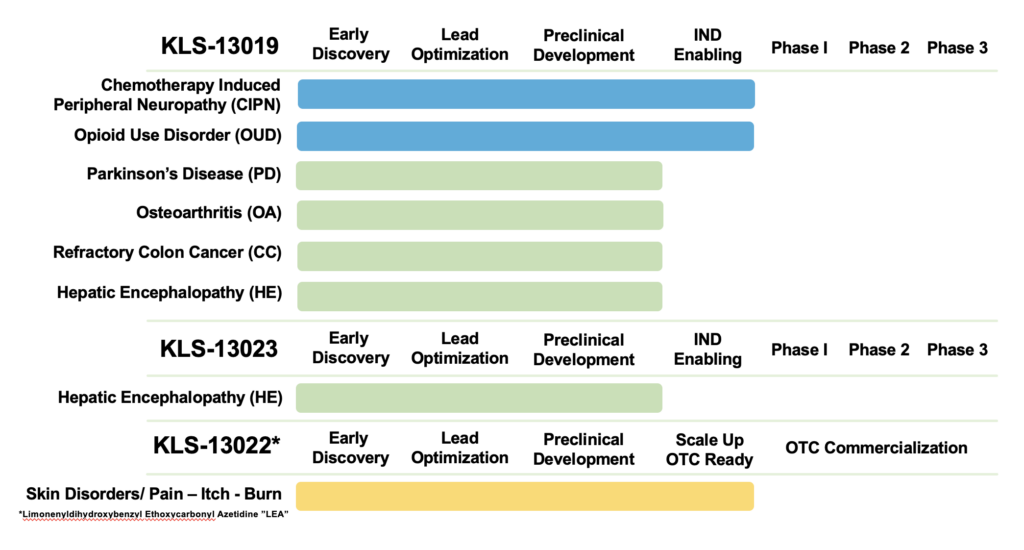
KLS-13019 (Oral Gelcap)
- Chemotherapy-Induced Peripheral Neuropathy (CIPN): Lead indication. Proven to prevent and reverse neuropathic pain in animal models. IND filing anticipated in 2026.
- Opioid Use Disorder (OUD): Shown to reduce opioid cravings, withdrawal symptoms, and anti-nociceptive tolerance in animals.
- Parkinson’s Disease (PD): Targeted to mitigate neuroinflammation and preserve neuronal cell health; studies currently underway.
- Refractory Colon Cancer (CC): Shown to significantly decrease tumor growth both alone and in combination with paclitaxel without negative interactions.
KLS-13023 (Oral Gelcap)
- Hepatic Encephalopathy (HE): Designed to target neurotoxicity caused by ammonia and ethanol in the brain. KLS-13019 has shown to be 50x more potent than CBD in protecting hippocampal neurons from oxidative stress associated with liver failure.
KLS-13022 (Topical Cream)
- OTC Skin Disorders: Target indications include pain, itch, and burn.
- Current Status: Early discovery/optimization, with a target to be OTC ready by late 2026.
Key Scientific Validations
- Animal Efficacy: Successfully demonstrated reversal of acute CIPN and prevention of neuropathic pain in animal models.
- Safety Profile: Clean toxicology profile in two species (rats and dogs) and demonstrated low abuse liability with no significant CNS behavioral differences from vehicle groups.
- Grant Funding: Research supported by $3.3M from the NIH HEAL Initiative and $1.5M from the Michael J. Fox Foundation.
- Peer-Reviewed Research: Supported by 9 peer-reviewed publications from 2016 to 2025.
- (1) ACS Medicinal Chemistry Letters (2016, 7, 424-428) “Discovery of KLS-13019, a Cannabidiol-Derived Neuroprotective Agent, with Improved Potency, Safety, and Permeability”
- (2) Journal of Molecular Neuroscience (14 August 2018) “Pharmacological Comparisons Between Cannabidiol and KLS-13019.”
- (3) Journal of Molecular Neuroscience (10 May 2019) “Knockdown siRNA Targeting the Mitochondrial Sodium-Calcium Exchanger-1 Inhibits the Protective Effects of Two Cannabinoids Against Acute Paclitaxel Toxicity.”
- (4) British Journal of Pharmacology (10 May 2019) “Behavioural and pharmacological effects of cannabidiol (CBD) and the cannabidiol analogue KLS-13019 in mouse models of pain and reinforcement.”
- (5) Journal of Molecular Neuroscience (02 July 2022) “Anti-Inflammatory Properties of KLS-13019: a Novel GPR55 Antagonist for Dorsal Root Ganglion and Hippocampal Cultures.”
- (6) Tetrahedron Letters (06 March 2023) “Efficient Syntheses of KLS-13019 Using Palladium Mediated Cross Couplings.”
- (7) Journal of Pharmacology and Experimental Therapeutics (June 2023) “GPR55 Antagonist KLS-13019 Reverses Chemotherapy-Induced Peripheral Neuropathy (CIPN) in Rats.”
- (8) Journal of Molecular Neuroscience (11 April 2024) “Knockdown siRNA Targeting GPR55 Reveals Significant Differences Between the Anti‑inflammatory Actions of KLS‑13019 and Cannabidiol”
- (9) Journal of Molecular Neuroscience (14 March 2025) “Effect of Fatty Acyl Composition for Lysophosphatidylinositol on Neuroinflammatory Responses in Primary Neuronal Cultures.”
Pipeline
We are in the leadership role as a commercial drug discovery company advancing cannabinoid therapeutics to successfully synthesize CBD derived new chemical entities (NCEs).
Intellectual Property
We have synthesized, preclincally tested and patented our proprietary CBD derived new chemical entities (NCEs), including KLS-13019 and also formulated a new CBD inspired molecule, KLS-13023. KLS-13019 is our lead target drug candidate and is part of an estate of NCEs underlying U.S. Patent 9,611,213 titled “Functionalized 1,3 Benzene-diols and their Method of Use for the Treatment of Hepatic Encephalopathy”. This patent is part of a divisional patent application by the Company to the USPTO whereby the Company sought separate claims for composition of matter, covered in Pat. 9,611,213 and separate claims for method of treatment; and U.S. Patent 10,004,722 titled “Method for Treating Hepatic Encephalopathy or a Disease Associated with Free Radical Mediate Stress and Oxidative Stress with Novel Functionalized 1,3 Benzene-diols.”